New ways to monitor offshore environments
A look at four novel methods and their advantages
Benjamin M. Kampala
Total E&P
Recent progress in techniques to monitor regular and planned exploration and production discharges offshore is expanding environmental management options for E&P companies. New water column and sediment measurement methods help make possible informed environmental management decisions. Such monitoring methods can be particularly important as E&P companies look to work in sensitive and previously unexplored environments that test the limits of conventional monitoring.
In some cases, tried and true methods have only limited applicability in deepwater operations and arctic projects. Furthermore, emissions from long-term, regular discharges are the subject of increased focus in terms of effects in the sea and in application of the best available treatment technologies.
Marine environmental monitoring can apply to permits and licenses, validation of numerical models, regulatory reporting, and technology selection. Nearly all the environmental management of an offshore installation relies in some way on the data from marine environmental surveys.
For example, the initial state of the seas surrounding a development are monitored for baseline data and, following start-up, monitoring of the sediment and water column is performed periodically to help ensure the good environmental condition. Also, technology selection can be validated, as in the case of a platform in Norway where water treatment engineers used a fish biomarker survey to demonstrate the effectiveness of improved produced water treatment.
Therefore, good, reliable data that represent temporal and spatial variation are needed to meet these and other environmental management needs. However, monitoring in the marine medium is challenging and limitations often restrict the amount of data available.
A challenging activity
Environmental monitoring around offshore E&P activities is expensive compared to the equivalent for land based activities. This means monitoring typically yields fewer samples and is performed less often. The principle contributor to cost is logistics, including a vessel from which to conduct activities. Shipping costs to offshore installations, transport, and analytical costs also push up the expense of marine environmental monitoring.
Spatial and temporal heterogeneity of the water column and seabed makes statistical significance of the data and results coming from these studies a challenge. Often the interpretation of monitoring data must rely on observed trends rather than statistically significant datasets. Consider that water column monitoring from a single sampling point may yield entirely different result on consecutive days, merely from a change in direction of ocean current.
Finally, it is not just cost and variability of the sampling zone that creates challenges. Rough seas, deep waters, arctic conditions, difficulty in sampling around an operating platform, bottom hazards such as pipes and risers, all combine to make environmental monitoring in the marine environment a planning challenge. Occasionally, unanticipated delays or errors caused by these complex situations could mean data is lost or costs rapidly increase.
Current monitoring methods
Conventional methods of water, benthic sediment, and benthic invertebrate sampling (the conventional sampling methods) are the workhorses of environmental sampling both offshore and onshore (lakes and rivers). They generally are robust and are considered valid by regulators and stakeholders. These monitoring techniques measure concentrations of substances associated with anthropogenic discharges, including PAHs, BTEX, nutrients, salts, and more.
The analysis of water and sediment samples provide data against which indices or guidelines may be compared, and also can be interpreted by biologists to give an idea of the functioning of the ecosystem and indications of perturbation.
The conventional approach of benthic invertebrate sampling provides data for community structure indices used to interpret ecosystem function. Indices such as Shannon's or density can reveal nutrient deficiency or enrichment. The principle drawbacks are that these methods are costly, time consuming, and do not indicate the short-term response, but rather the response from years of exposure.
A need for new methods
The use of conventional sampling methods at sea persists partly due to the good data they provide, but also in the case of water column analyses, no alternatives have been available until recently.
With changing regulatory, technical, and other data needs, elaborated methods are needed, particularly in new environments like arctic and deep offshore. They should be cheaper and easier to apply. They should provide additional figures against which indices or guidelines may be measured. They also may provide new types of data, such as information about the ecosystem's condition. To meet these requirements, new methods have been developed and are starting to see wider application.
When attempting to use a new method, advance testing and study are required and may include a bibliography, lab testing, and pilot studies. The parameters to be reported should be well understood and quantifiable to known limits of detection and uncertainty. Equally important are the spatial and temporal time scales for which the data will be considered valid. Whether results are for physical or chemical parameters should be clear as well as their significance to the ecosystem. Finally, it should be understood which environmental compartment results are indicative (water or sediment).
Once a method is well known, the preferred method to pilot a study is a comparative test. The concurrent testing of monitoring methods permits direct comparison of results and thus validation of a method.
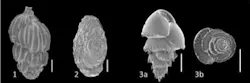
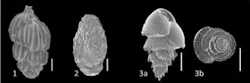
Foraminiferal assessment
Foraminiferal assessment and ecotoxicological testing offer alternatives to conventional sediment compartment monitoring.
Foraminifera are unicellular protists with a calcareous shell. To conduct a foraminiferal assessment, samples of sediment must be taken at each station. Once aboard the vessel, the samples are preserved and the topmost layer of sediment is retained for analysis. Laboratory analysis consists of sorting, identifying, and counting the individuals found in a given sieve size.
The benefit of foraminiferal assessment over conventional analyses is that one sample offers data from the period prior to operational activities, as well as indications of effects from drilling discharges. The presence of fossil assemblages permits interpretation of historical conditions, while living foraminifera permit interpretation of current conditions. Analyses can be done on small sample volumes, and studies may offer improved statistical representivity of the area. Moreover, the turnover of organisms is faster than benthic sediment and benthic invertebrates, giving a faster response to environmental conditions. Another argument for foraminiferal assessment is that it can be used to sample in extreme conditions (deepwater, arctic environments) where benthic invertebrates are often not present in large numbers.
The analysis is completed by applying the counted individuals, both living at the time of sampling (identifiable by the preserving agent rose Bengal in tissues) and those from deeper layers of sediment which do not absorb the rose Bengal, and thus are known to be dead at the time of sampling. Indices of community structure and trophic function give insight into the presence of opportunist species and those species sensitive to the substances in the drilling discharges. A similar proportion of each species of foraminifera in both the living and deeper, dead fractions, as well as homogenous community structure, indicates no effect from anthropogenic discharges.
The cost of a foraminiferal assessment is roughly the same as a benthic invertebrate assessment, but foraminifera yield data in deepwater and historical data, making it a more cost-effective alternative in certain situations. Whereas foraminiferal assessment is analogous to the techniques and analysis of benthic invertebrates and similar in costs, ecotoxicological testing is different and may be done at lower costs.
Ecotoxicological testing
Ecotoxicological testing is not new. What is novel is conducting the tests in an easy-to-use manner on sediments in contact with drilling discharges. A feature of ecotoxicological testing is that the sediment samples are tested in a relevant environmental medium on species with ecological relevance. The test gives an idea of disruption to the ecosystem as a whole rather than just the sum of measured chemicals (which leaves out synergistic effects and effects of those compounds not measured).
Ecotoxicity testing of sediment is done by suspending sediment in clean water and adding reference larvae. The presence of altered larval development stages when examined after 24 hours of incubation is the measure of toxicity and relates to the presence of xenobiotics in tested samples. In effect, the toxic response of the organism can be interpreted to give an indication of effect from anthropogenic discharges. An absence of toxicity indications can be interpreted as no effects. The practical and logistical requirements are equivalent to conventional methods, but ecotoxicological testing can be done at no additional cost or complication to a sampling program. Sampling is easy and can be performed wherever benthic sediment samples are collected, which enables a practical evaluation of spatial extension of toxicity. This provides good information at a low cost.
Passive samplers
Another way to sample concentrations in the water column may provide a number of practical and logistical advantages. Passive samplers permit collection of a time-averaged sample of constituents present in the water column. The sample is gathered slowly over several days, weeks, or months. While conventional water column analyses may have low detection limits, a large volume of water would have to be filtered to find adequate concentrations. This is difficult, and even if realized, the results of a high-volume conventional approach would represent only the short time period where water is being pumped and filtered. With passive samplers, the substances that pass through the membrane and integrate into the sampling medium are considered to represent a time averaged concentration for the entire period of immersion (weeks or months). As such, results from passive samplers have good representation of average concentrations.
A passive sampler being deployed at sea. Passive samplers are used to sample substances in the water over a period of weeks and are capable of measuring PAHs, BTEX, metals, and other substances. (Photo courtesy Total EP)
When an array of passive samplers is deployed around a platform, results can be presented as isopleths of concentrations on a chart. Passive samplers can measure polyaromatic hydrocarbons (PAHs), benzene/toluene/ethylbenzene/xylene (BTEX), metals, and other substances. This can be cost effective. The cost of passive samplers is about the same as for conventional analyses, yet they yield considerably more data. The deployment and retrieval of such samplers may be performed using small craft such as "surfers." A principle advantage of this method is the eventual ability to provide datasets to validate produced water dispersion models.
Biomarkers
The biomarker approach has advantages that are quite different than the previous methods. Conducting an analysis of biomarkers involves testing tissue samples for physiological changes that occur uniquely as a result of exposure to a given anthropogenic substance – in this case those present in produced water discharges. The biomarker concept works on the premise that observed physiological or molecular changes in marker species, when compared to reference areas, can be interpreted to explain effects on marker species from emissions.
The timescale of response suits recent discharges (hours/days/weeks) rather than the long leads needed for some other methods. The result is a short-term, local adaptation or response of an individual, rather than a long-term population response in the ecosystem.
Sampling methods for biomarker assessment vary depending on study design and the species used as markers. Gathering for tissue samples can be done by caging of fish and mussles, fish traps, or conventional fishing. Tissue samples may include bile, gills, blood or other, and analysis is performed with a high level of analytical precision. Depending on the experimental design of a given biomarker study, results can be interpreted on diverse spatial and temporal timescales and may include considerations about the fish migration and exposure to discharged material.
The principle drawback of the biomarker method is caused by the uncertainty in results arising from confounding factors. The complexities of ecosystems offer many possible sources to activate biomarkers, potentially altering interpretation. However, when properly planned, it is possible to discern biomarker effects resulting from E&P activities and background effects.
The biomarker method has the ability to overcome uncertainty and in doing so, be an example of an application of evidence-based monitoring. Most monitoring methods require an understanding of pathway and causes resulting in a signal being interpreted. Biomarkers accept that pathways between anthropogenic substances and organisms are complex and simply provide a measure of exposure. Thus, a well designed experiment can demonstrate relative levels of exposure by organisms around an offshore platform, but avoid the need for complicated interpretation. No direct causality needs to be established for biomarkers.
The types of analyses that are typically performed include EROD (ethoxyresorufin-O-deethylase) which is a measure of PAH detoxification, analysis for by-products of PAH metabolism, and histopathology. While each of these analyses provides an indication of an effect on tissue, knowing the exact type of PAH that causes the signal is not needed. This means that the method is robust to detect several pathways of effects without needing to know exactly what they are or how anthropogenic substances act on the tissue (and possibly interact with natural substances). Development of these methods has come from several years of study and collaboration among scientists, industry, and regulators.
Ongoing research needed
Further research is needed to improve ease and function of biomarker, as well as other monitoring methods. The monitoring methods presented here exist at different stages of development. Regardless of the stage, ongoing R&D is needed to innovate and continue to prove the effectiveness of environmental monitoring. This may include the application of existing monitoring methods and the new methods together in order to achieve an integrated understanding of the environment. By doing so, the benefits of these techniques and analytical methods can find new applications and be more readily applied. Such integrated understanding could then indicate what should be a most informative and cost efficient approach to regular offshore monitoring in the future.
Also, the changing business needs which spurred development of these novel methods persist. As oil and gas production evolves, so too will the needs and stakes which drive research and investment in new methods. Those methods which respond best to changing stakes should be supported and R&D efforts should preferentially be invested.
New methods offer rigorous and useful alternatives. Already from using foraminiferal assessments, passive samplers, ecotoxicology analysis, and biomarkers, it is clear that important gains have been made in terms of cost effectiveness, spatial and temporal coverage, geographic applicability, and analytical abilities.
Acknowledgment
The author wishes to thank Jan Fredrik Børseth of IRIS Biomiljø and Francois Galgani of IFREMER for providing comments and input to this document.
Offshore Articles Archives
View Oil and Gas Articles on PennEnergy.com